|
4/12/2024 0 Comments Valence electrons periodic table 
In practice, chemists simplify the notation by using a bracketed noble gas symbol to represent the configuration of the noble gas from the preceding row because all the orbitals in a noble gas are filled. As we proceed downwards in a group, the numbers of valence electrons are same, although the number of shells increases. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. The number of valence electrons in an atom is reflected by its position in the periodic table of the elements (see the periodic table in the Figure below). Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. Another common method of categorization recognizes nine element families: Alkali Metals: Group 1 (IA) - 1 valence electron. The increasing positive charge attracts the electrons more strongly, pulling them closer to the nucleus. Here, we just refer to the periodic table and search for the position of the element in it. Valence Electrons and the Periodic Table. As we go across a period on the periodic table, however, electrons are being added to the same valence shell meanwhile, more protons are being added to the nucleus, so the positive charge of the nucleus is increasing. Atoms share electrons and form covalent bonds to satisfy the octet rule. This page titled 5.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by Anonymous. The atoms in group 6A make two covalent bonds. 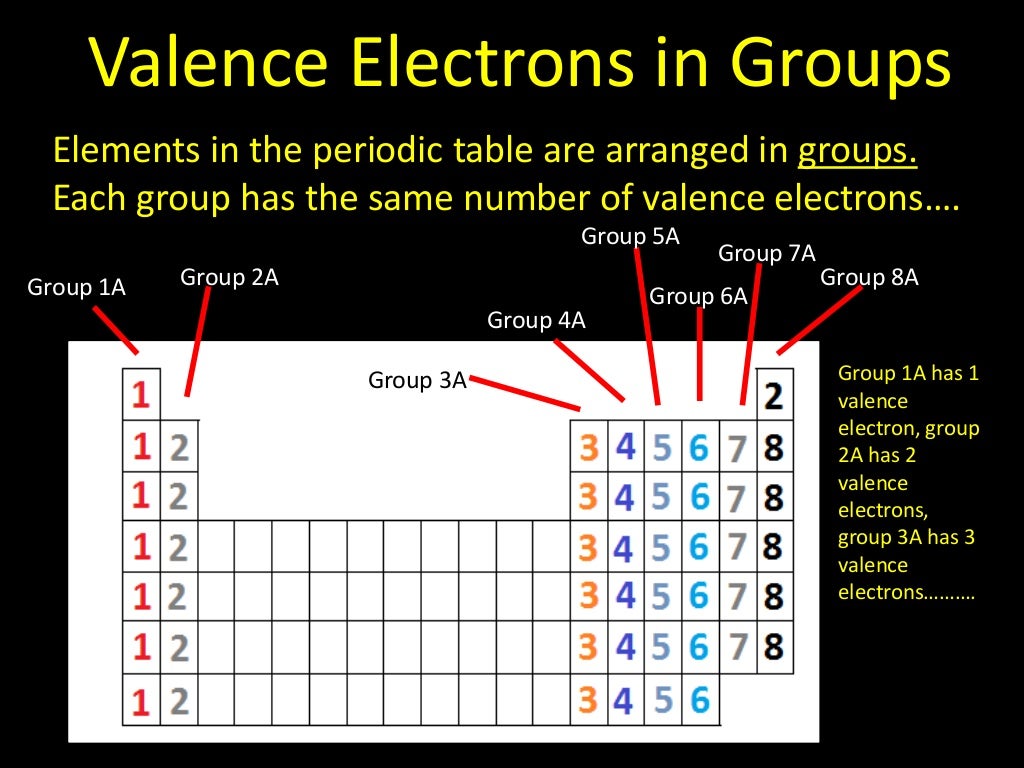
When we reach neon, with Z = 10, we have filled the 2 p subshell, giving a 1 s 22 s 22 p 6 electron configuration:Īs we continue through the periodic table in this way, writing the electron configurations of larger and larger atoms, it becomes tedious to keep copying the configurations of the filled inner subshells. It is the most widely used method to determine the number of valence electrons in an element. Covalent bonds are formed by two atoms sharing electrons. How many valence electrons does an element have You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of the element) has. Similarly, fluorine has the electron configuration 1 s 22 s 22 p 5: Fluorine has one electron pair in the 1 s orbital, one electron pair in the 2 s orbital, and 2 electrons pairs with one unpaired electron in the 2 p orbital. On the other hand, nitrogen can form NH 3 so it has a valence of 3, and 3 valence electrons. 8.4: Electron Configurations, Valence Electrons, and the Periodic Table. Oxygen has one electron pair in the 1 s orbital, one electron pair in the 2 s pair, and one electron pair, and 2 unpaired electrons in the 2 p orbitals. Map: A Molecular Approach (Tro) 8: Periodic Properties of the Elements. See examples, diagrams, and tips for this chemistry concept. Because all the 2 p orbitals are degenerate, it doesn’t matter which one has the pair of electrons. Learn how to identify the number of valence electrons for main group elements on the periodic table based on their group number. One electron must be paired with another in one of the 2 p orbitals, which gives us two unpaired electrons and a 1 s 22 s 22 p 4 electron configuration. 
5.17: Electron Configurations and the Periodic Table. The electron configuration of nitrogen is thus 1 s 22 s 22 p 3.Īt oxygen, with Z = 8 and eight electrons, we have no choice. ChemPRIME (Moore et al.) 5: The Electronic Structure of Atoms. The number of valence electrons depends on the octet rule. Elements within the same group share the same number of valence electrons. Atoms in a period have the same number of electron shells. An interactive periodic table may be found here.\): Copy and Paste Caption here. Valence Electron BasicsLearn how to use the periodic table in order to determine the number of valence electrons.The valence electrons are the electrons foun. An element period is a horizontal row on the periodic table. \): The modern version of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
